- Home Page
- Company Profile
-
Our Products
- Laboratory Chemicals
- Ferrous Sulphate Heptahydrate Extrapure ACS 99
- 3-Amino-1-propanol For Synthesis
- Silver Sulphate 99.5% AR/ACS
- Nitric Acid 2.5L Honeywell
- Honeywell Hydranal Coulomat AG
- Sodium Thiosulphate ER 500g
- Whatman High-Purity Glass Soxhlet Extraction Thimbles
- Sodium Acetate Trihydrate Ar
- Rankem Benzaldehyde Reagent
- 7782-63-0 Extrapure Ferrous Sulphate Heptahydrate
- HYDRANAL Coulomat AK Honeywell
- 100G ER Silver Nitrate
- Honeywell Ethanol 500ML
- 1327-53-3 Arsenic Trioxide
- 54-11-5 Nico tine For Synthesis
- 250ML Conductivity Standard Hamilton
- 64-17-5 500ML Honeywell Ethanol
- 7647-14-5 SQ Sodium Chloride
- 7757-83-7 Sodium Sulphite Anhydrous
- 108-88-3 Sulphur Free Tol uene
- 71432 99.5 ACS Benzene
- 1762-95-4 Ammonium Thiocyanate Crystal
- 7631-99-4 SQ Sodium Nitrate
- 10294-42-5 Ceric Sulphate Tetrahydrate extrapure AR
- 7783-20-2 ER ACS Ammonium Sulphate
- 10043-52-4 SQ Calcium Chloride Dihydrate
- 6381-92-6 EDTA Disodium Salt Dihydrate extrapure AR
- 631-61-8 ER ACS Ammonium Acetate
- 7757-82-6 Sodium Sulphate Anhydrous
- L Ascorbic Acid SQ 500
- HYDRAZINE SULPHATE ACS, REAG. PH.EUR. UR"UNIVERSAL REAGENT" PASSES PURE & AR
- Plate Count Agar Standard Methods Agar
- Labolene Neutral 500ML
- Virosil Pharma 5LTR
- Phospho Histone H2A.X(Ser139) Monoclonal Antibody(3F2) , Packaging Type: Bottle, 1 ml
- Ammonia Solution 25% SQ 500ML
- Sterind Bowie And Dick Test Pack 121 Deg 15 Mins
- Titration Dpd No 4 Tablets
- N-Methyl-2-Pyrrolidone (NMP) extrapure AR, 99.5
- Qualigens Acetonitrile Chemical
- Hydroxylamine Hydrochloride extrapure AR
- FLUORESCEIN 95%
- AMMONIUM ACETATE CRYSTALS EP , 500gm Bottle
- N Heptane ER 500ML
- Hydrochloric Acid Honeywell
- Rapid Refined Vegetables oil Adulteration Test Kit 50 Tests
- Nddb Milk Adulteration Test Kit Large Size
- SODIUM TRIACETOXY BOROHYDRIDE
- CHROMIUM AAS STANDARD SOLUTION 1000 mg /
- SODIUM BOROHYDRIDE Extra Pure
- PHOSPHORUS AAS STANDARD SOLUTION
- CURCUMIN CRYSTALLINE Extra Pure
- N 1 NAPHTHYL ETHYLENE DIAMINE DIHYDROCHLORIDE AR RIDE AR, Analytical Grade, 98%
- ACID Extra Pure
- BEEF EXTRACT POWDER Bacteriological grade 500G
- ETHYLENEDIAMINE TETRAACETIC ACID MAGNESI
- SULPHANILAMIDE Extra Pure
- MERCURIC SULPHATE AR
- BARIUM DIPHENYLAMINE SULPHONATE AR
- Ph Buffer Solution
- N-1-NAPHTHYL ETHYLENE DIAMINE
- POTASSIUM PERMANGANATE
- FLUORESCEIN 99.0%
- LABDET 05 (NEUTRAL) PHOSPHATE FREE
- MAGNESIUM SULPHATE HEPTAHYDRATE AR, ACS
- FAST GARNET GBC SALT
- FAST GREEN FCF
- FAST GREEN FCF
- FAST GREEN FCF
- Sodium 1-pentanesulfonate monohydrate
- Chlorotex Reagent 100ML
- Calcium Hardness Tablets
- Amoxicillin AMX
- TSB Tryptone Soya Broth Supplemented
- Chlorotex Reagent 100ML
- Dichloran Rose Bengal Agar (DRB
- Violet Red Bile Agar 500G
- For Laboratory Hcl Hydrochloric Acid
- Merck Emplura 2.5L
- Whatman 1822-025 Grade GF/C Glass Microfiber Filter Papers 1.2 um 25 mm
- TDS 442 tandard Solutions 50ppm
- Dpd Tablets No 1 for water testin
- TDS 442 tandard Solutions 300ppm
- Hydrochloric Acid 08256 6, Packaging Details: Plastic Bottle
- Hepes Buffer Extrapure, 99
- Ammonium Persulfate
- Indicator Papers Full Range Qualigens
- Paraffin Liquid Light
- Hepes Buffer Extrapure, 99
- Ferrous Sulphate Heptahydrate Reagent
- Hamilton Conductivity Standard 13, 250ML
- Stannous Chloride SQ 100G ,
- Hexane ER 500ML
- Potassium Fluoride Anhydrous SQ 500G
- Bottle Starch Soluble ER 500G
- Nitric Acid ER 500ML , Packaging Details: Glass Bottle
- Powder Ammonium Persulfate
- SALICYLIC ACID AR COMPLIES IP
- Hydrogen Peroxide Solution ER 500 ML
- Sodium tetraborate decahydrate
- MAGNESIUM SULPHATE HEPTAHYDRATE AR, ACS COMPLIES IP, BP, USP, PH.EUR , Packaging Details: Bottle
- MAGNESIUM CHLORIDE HEXAHYDRATE AR COMPLIES IP, BP, USP, PH.EUR , Packaging Details: Bottle
- Powder Ammonium Persulfate , Grade Standard:
- EDTA Disodium Salt SQ 500G , Packaging Details:
- Sodium Hydroxide Pellets SQ 500G
- n Hexane For HPLC
- Isooctane For Analysis EMSURE ACS, Reag. Ph E
- Residual Free Chlorine (RFC)
- FAST GREEN FCF For Microsco
- Spectroquant Lead Test Range
- Boric Acid Crystals 500 Gm
- Powder Ammonium Per Sulphate (SQ), Qualigens
- POTASSIUM PERMANGANATE AR, ACS COMPLIES
- Barium Chloride ER 500G , For
- Qualigens Oxalic Acid SQ 500G
- Oxalic Acid ER 500G
- Calcium Chloride Dihydrate
- Tartaric Acid SQ 500G
- Ph Bromothymol Blue Blue Indicator
- iso Propyl Alcohol SQ , Packaging Details: Plastic
- Ammonia Buffer Solution 500ML , Packaging Detai
- Phenolphathalin Indicator Solution
- Electrode Storage Solution (500 ML) - HI70300L
- 3.5M KCl Electrolyte Fill Solution For Double-Junc
- SODIUM CITRATE SOLUTION 3.8% w/v
- DI-SODIUM HYDROGEN ORTHPHOSPHATE ANHY
- PARAFFIN LIQUID
- BROMINE WATER APPROX. 0.2 M
- Silica Gel Self Ind Coarse SQ 500G
- METHYL VIOLET STAINING SOLUTION For Microsc
- MANGANESE AAS STANDARD SOLUTION
- LITHIUM AAS STANDARD SOLUTION 1000 mg/
- Chemical Grade Powder Ammonium Molybdate ER
- Silver Nitrate Acs 25G ackaging Details: Bottle
- Ammonium Dihydrogen Ophosphate SQ , Packaging Details: Bottle
- METHYLENE BLUE SOLUTION For Microscopy
- GLUCOSE STANDARD STOCK
- L Ascorbic Acid , Packaging Details: 500GM
- Potassium Hydroxide Pellets
- Potassium Iodide Chemical , 99%
- SODIUM SILICATE (META) NONAHYDRATE 500G (
- HYDROXYLAMMONIUM HYDROCHLORIDE AR, ACS
- Silver Nitrate ER 100G
- NITRIC ACID AR
- HEXANE AR (ALL SATURATED ACYCLIC HYDROCA
- Sodium Acetate Anhydrous SQ 500G
- ORTHOPHOSPHORIC ACID SP GR 1.75 AR 2.5L
- ORTHOPHOSPHORIC ACID SP.GR.1.75 AR (phosph
- Potassium Iodate SQ 250G
- CURCUMINE CRYSTALLINE
- METHYL ORANGE (PH INDICATOR)
- SODIUM THIOSULPHATE PENTAHYDRATE AR, ACS, ISO 500G
- POTASSIUM PERMANGANATE
- FAST GREEN FCF For Microscopy
- Arsenic Trioxide 99% AR
- MERCURIC SULPHATE AR
- Qualigens Di Sodium Tetraborate
- Ph Buffer Solution
- Diiodomethane .
- MANGANESE AAS STANDARD SOLUTION 1000 m
- LITHIUM AAS STANDARD SOLUTION 1000 mg/L Li
- GLUCOSE STANDARD STOCK
- AMMONIUM CITRATE 10% w/w SOLUTION
- NITROSO-R-SALT AR
- HYDROXY NAPHTHOL BLUE Indicator grade LOBA
- STANNOUS CHLORIDE DIHYDRATE 98% AR/ACS
- POTASSIUM FERRICYANIDE 98% Extra Pure
- TETRACHLOROETHYLENE 99% Extra Pure
- Brilliant Blue G 250 See COO
- ERYTHROSINE B 80% For Microscopy
- POTASSIUM OXALATE MONOHYDRATE 99% Extra
- CARMOISINE A 50% dye content Extra
- TARTRAZINE AR/For Microscopy
- TRICRESYL PHOSPHATE Extra Pure
- Exeter Campylobacter Selective Broth Base
- Brilliant Blue G 250 See COOM
- CARBOL FUCHSIN DILUTE STAINING SOLUTION
- Tris (hydroxymethyl) aminomethane for analysis , Packaging Details: 500G
- LABDET 05 NEUTRAL PHOSPHATE FREE
- FLUORESCEIN, 25G
- Zinc Oxide SQ 500G
- O Cresoal Solvent 500ML LOBA
- Charcoal Activated Granular
- CHARCOAL ACTIVATED PHOSPHORUS FREE AR
- Zinc Oxide SQ 500G
- Calcium Carbonate
- Butylated Hydroxy Anisole , Packaging Details: Pl
- Mercuric Chloride 100g
- Mercuric Chloride 100g
- Cellulose Powder , Packaging Type: Bottle, Packag
- Sodium Carbonate Anhydrous
- Mayer Solution
- SORBIC ACID 99% Extra Pure
- Sulphuric Acid SQ 5L
- Distilled Water SQ 5L
- Ethyl Acetate SQ 2.5L
- Litmus Blue Indicator Papers 10Booklets
- Rankem Liquid Paraffin Light , 100%,Rankem Liquid Paraffin Light , 100%,Rankem Liquid Paraffin Light , 100%,
- Phenol (Carbolic Acid) SQ 500G
- Qualigens Sulphuric Acid SQ
- Di-Ammonium Hydrogen O
- Rankem Liquid Paraffin Light , 100%,
- Ammonium Acetate SQ 500G
- Grade: Chemical Grade Copper Sulphate Pentahyd
- Benzene Crystallizable Reagent
- 500g Ferric Chloride Anhydrous SQ
- Glass REAGENT BOTTLES Narrow Mouth With Screw Cap, Amber , 1000 ml
- Ethanol Ar China Ethyl Alcohol
- Potassium Dihydrogen Orthophosphate extrapure A
- Calcium Sulphate Dihydrate
- Tetraethyl Orthosilicate (TEOS) extrapure, 98%, 28
- Ammonium Sulphate For Analysis Emsure ACS, IS
- N-HEXANE EMPLURA 2,5 L
- Iodine Solution N 10
- P H Indicator Paper Universal 2.0 - 4.5
- Salmonella H Antiserum Poly a-z 3mL BD-Difco
- Liquid Sulphuric Acid
- 40% Qualigens Hydrofluoric Acid 48% HF , For Labo
- Methyl Red Indicator Solution 125ML
- Qualigens Ammonium Persulphate
- Sulfanilic Acid 25G
- Potato Starch pure, For Food
- Perchloric Acid 70% ER
- Buffer Tablets PH 9.2 , Grade Standard: Analytical Grade
- 99% Benzene EMPLURA MERCK, Chemical Grade, 500ml
- Metanil Yellow
- Caspofungin CAS 5 mcg
- Ampicillin Sulbactam 10 mcg
- Methanesulphonyl Chloride (MSCl) pure, 98%
- n-Heptane for HPLC & UV Spectroscopy, 99.5%
- Qualigens Phenolphthalein Solution
- Ethanol 99.99
- Paraffin Liquid Light
- Hydrochloric Acid SQ 500ML
- Ozone Test Kit Dpd Tablet No 4
- Hexane Sulphonic Acid Sodium Salt Anhydrous 99% , 25G, AR (Analytical)
- Sodium Hydroxide Solution
- Acetic Acid Glacial SQ 2.5l
- Lactophenol Cotton Blue
- Arsenic Trioxide 99% AR Loba
- Carbol Fuchsin Dilute Staining Solution Loba
- Methanol Hplc Gradient Grade Qualigens
- Water HPLC Qualigens
- D-(+)-Xylose Himedia
- Ammonia Solution AR/ACS Loba
- 1 Hexane Sulphonic Acid Sodium Salt Anhydrous 99%
- 35M KCl Electrolyte Fill Solution For DoubleJunction Electrodes 4 X 30mL HI7082
- 98% Sodium Arsenite
- 250Ml Hamilton Conductivity Standard 13
- 500ml Triethanolamine SQ
- Absolute Ethyl Alcohol 500ml
- Acetanilide Powder 98% Extra Pure
- Acetic Acid Glacial SQ 2.5l
- Acetone SQ 500ml Qualigens
- Fraser Broth Base Himedia
- Adipic Acid SRL
- ALUMINIUM CHLORIDE HEXAHYDRATE 99% AR
- Aluminium Chloride Hexahydrate Extrapure AR 99% SRL
- Ammonia Buffer Solution 500ml
- Ammonia buffer solution
- Ammonium Acetate Crystals EP
- Ammonium Acetate ER ACS 500gm Qualigens
- Ammonium Acetate SQ 500g
- Ammonium Carbonate Qualigens
- Ammonium Dihydrogen Orthophosphate Qualigens
- Ammonium Ferrous Sulphate ER 500g Qualigens
- Ammonium Iron II Sulfate Hexahydrate (2)
- Ammonium Iron II Sulfate Hexahydrate
- Ammonium Molybdate Rankem
- Ammonium Molybdate SQ
- Ammonium Molybdate Tetrahydrate ER 100g Qualigens
- Ammonium Per Sulphate SQ 500g
- Ammonium Persulphate Qualigens
- Ammonium Phosphate Dibasic 500g
- Ammonium Phosphate Monobasic Extrapure AR, 99%
- Ammonium Sulphate ER ACS
- Ammonium Thiocyanate Crystal 500gm
- Ammonium Thiocyanate Crystals SQ 500gm
- Arsenic Trioxide 99% AR LOBA (2)
- Arsenic Trioxide 99% AR Loba
- Asparagine Proline Broth, Granulated
- Baird Parker Agar Base Himedia
- Barium Chloride Dihydrate
- Barium Diphenyl Sufonate SQ 25g Qualigens
- Barium Diphenylamine Sulphonate AR
- Benzene ACS 995
- Benzene Crystallizable Reagent
- Benzene EMPLURA MERCK
- Bleaching Powder Extra Pure LOBA
- Borom Trifluoride Ethyl Ether Complex
- Brilliant Blue G for Electrophoresis Qualigens
- Brilliant Blueg 250 See Coomassie Brilliant Blue G 250
- Brilliant Green Agar Base, Modified
- Bromine Water Approx 0.2m
- Bromophenol Blue Indicator
- Bromothymol Blue Blue Indicator
- Bromothymol Blue Indicator Solution
- Buffer capsules pH 9.20 Merck
- Buffer Solution Tr Merck Ph 10
- Buffer Tablets PH 9.2 Qualigens
- Butylated Hydroxy Anisole LOBA
- Butylated Hydroxytoluene Powder
- CADMIUM ACETATE DIHYDRATE 98% Extra Pure LOBA
- Calcium AAS Standard Solution 1000mg
- Calcium Carbonate SQ 500g Qualigens
- Calcium Chloride Dihydrate SQ 500gm
- Calcium Chloride Dihydrate SQ
- Calcium Chloride Dihydrate
- Calcium Hydroxide SQ
- Carbol Fuchsin Dilute Staining Solution Loba
- Cellulose Powder Loba
- Ceric Sulphate Tetrahydrate Extrapure AR
- Charcoal Activated Granular
- Chloroacetyl Chloride 98 LOBA
- Chloroacetyl Chloride 98% LOBA
- Chloroform SQ
- Chromium Trioxide SQ 500gm Qualigens
- Citric Acid Anhydrous
- Crystal Gentian Violet
- Curcumin Crystalline Extra Pure
- Curcumine Crystalline Diferuloylmethane
- Dextrose ACS (2)
- Dextrose ACS
- Di Potassium Hydrogen Orthophosphate SQ 500G Qualigens
- Di Sodium Tetraborate ER
- Di-Ammonium Hydrogen Orthophosphate SQ
- Dibutyl Phthalate SQ 2.5ltr
- Dichloromethane MDC ACS 995
- Dichloromthane Stab Amlene LVCG GRADE 99.90%
- Diethyl Ether Solvent
- Diiodomethane 98 For Synthesis LOBA
- Diiodomethane For Synthesis LOBA
- DiSodium Hydrogen OPhosphate Dih SQ 500G Qualigens
- Dusting Powder Sulphanilamide
- EDTA 0.1M C V S Qualigens
- EDTA Disodium Salt Dihydrate Extrapure AR
- Electrode Storage Solution (500 ML) - HI70300L
- Emplura Tin Metal
- Eriochrome Black T C.I. 14645 Indicat
- Eriochrome Black T Indicator
- Ethanol absolute AR ACS for Analysis
- Ethanol Fisher
- Fast Green FCF For Microscopy
- Ferric Chloride Anhydrous SQ
- Glucose Broth Himedia
- Glycerol analysis Merck
- Glycerol SQ Qualigens
- Glycine ACS 99.5%
- Hayman Premium Grade Ethanol 100% (2)
- Hayman Premium Grade Ethanol 100%
- Hematoxylin Stain 80 For Microscopical Staining
- HEPES Buffer Extrapure 99%
- Heptane Sulphonic Acid Sodium Salt Anhydrous
- HiCrome Listeria Ottaviani Agosti Agar Base Himedia
- Honeywell Ethanol 500ml
- Hydrofluoric Acid 40%
- Hydrofluoric Acid 500ml Qualigens
- Hydrogen Peroxide solution 30 SQ Qualigens
- Hydroxy Naphthol Blue Indicator Grade Loba
- Hydroxyapatite Nanopowder SRL
- Hydroxylamine Hydrochloride 99 ArAcs LOBA
- Hydroxylamine Hydrochloride 99% ARACS
- Hydroxylamine Hydrochloride Extrapure AR 99%
- Hydroxylamine Hydrochloride Extrapure
- Hydroxylammonium Hydrochloride Ar Acs
- Hypersil MOS-2 C8 HPLC Columns
- Iodine Solution N 10
- Labdet 05 Neutral Phosphate Free
- Labnol Absolute Ethanol Analytical AR Grade
- Laboratory Reagents
- LB Agar Miller
- Lead Oxide Red Qualigens
- Lithium AAS Standard Solution 1000 MgL Li In Diluted HNO3 Traceable To NIST Loba
- Litmus Blue Indicator Papers 10Booklets Qualigens
- Litmus Red Indicator Papers 10 Booklets
- M Cresol Solvent
- Magnesium Chloride Hexahydrate
- Magnesium Sulphate Heptahydrate SQ
- Mayer Solution Loba
- Merck Emplura Acetone 2.5L
- Mercuric Chloride SQ 250gm
- Mercuric Sulphate 250gm
- Mercury II Chloride For Analysis EMPARTA ACS
- Methanol AR 2.5l Qualigens
- Methyl Orange Ph Indicator
- Mueller Hinton Agar
- Mueller Kauffman Tetrathionate Novobiocin Broth Base Himedia (2)
- Mueller Kauffman Tetrathionate Novobiocin Broth Base Himedia
- N Methyl 2 Pyrrolidone NMP ACS 99%
- N1Naphthyl Ethylene Diamine Dihydrochloride ARACS
- Nicotine SIGMA ALDRICH
- Nicotinic Acid Extra Pure
- Nitric Acid AR
- Nitric Acid SQ Qualigens
- Nitroso-R-Salt AR Loba
- NMethyl2Pyrrolidone NMP ACS 99
- O Cresoal Solvent 500ml Loba
- Orthophosphoric Acid SP.GR.1.75 AR Phosphoric acid AR 2.5lt
- Oxalic Acid SQ 500g Qualigens
- Patton Reeder s Reagent New 5g
- Ph Buffer Solution LOBA
- Phenolphthalein Solution 125ml Qualigens
- Phosphomolybdic Acid ER ACS
- Phosphorus Standard for ICP
- Polyethylene Glycol 4000 SQ Qualigens
- Potassium Carbonate 500gm
- Potassium Carbonate Tci
- Potassium Chloride EMPLURA Merck
- Potassium chloride EMPLURA
- Potassium Chromate ER ACS Qualigens
- Potassium Dichromate Emplura
- Potassium Ferricyanide 98% Extra Pure
- Potassium Ferrocyanide AR
- Potassium Fluoride Anhydrous SQ 500gm
- Potassium Hydrogen Phthalate Volumetric Standard Secondary Reference Material for Alkalimetry
- Potassium Hydroxide Pellets SQ 500g Qualigens
- Potassium Hydroxide Pellets SQ 500g
- Potassium Oxalate Monohydrate 99
- QuickRxn NotI 20ul
- Rankem Fehlings Solution A
- Safranin, 0.5% wv Himedia
- SELENIUM POWDER BLACK 99.9% trace metal basis AR
- Silica Gel Blue Self Indicating Coarse SQ Qualigens
- Silica Gel Self Ind Coarse SQ 500g
- Silver Nitrate N10 Solution
- Silver Nitrate N50 Solution 500ML
- Silver Sulphate SQ Qualigens
- Sodium Arsenite 98 AR Loba
- Sodium Carbonate Anhydous SQ 500g
- Sodium Carbonate Anhydrous LOBA
- Sodium Chloride for Analysis Emparata Merck
- Sodium Chloride SQ 500g Qualigens
- Sodium Citrate Solution 3.8% WV Loba
- Sodium Hydrogen Carbonate ER 500G Qualigens
- Sodium Nitrate SQ
- Sodium Silicate (Meta)Nonahydrate 500G
- Sodium Sulfite Anhydrous ER ACS Qualigens
- Sodium Sulphate Anhydrous 500gm
- Sodium Sulphite Anhydrous 500gm Qualigens
- Sodium Sulphite ER ACS Qualigens
- Sodium Thiosulphate SQ Qualigens
- Sodium Triacetoxy Borohydride
- Stannous Chloride Dihydrate 98 ARACS
- Starch Soluble SQ 500g Qualigens
- Sulphuric Acid 98% Emplura
- Sulphuric Acid SQ 5l Qualigens
- Sulphuric Acid SQ
- Tartaric Acid SQ 500gm
- Tetrabutylammonium Perchlorate 99%
- Tetrachloroethylene For Spectroscopy Uvasol
- Tin Metal Granular 100gm
- Toluene Sulphur Free
- Tricresyl Phosphate Extra Pure
- Triethanolamine 98 Extra Pure
- Triethylene Glycol Extrapure
- Tris Hydroxymethyl Aminomethane for Analysis
- Turmeric Indicator Papers
- Universal Indicator Solution Ph 4.0-11.0
- Universal Indicator Solution Ph 4.11 Qualigens
- Water For HPLC
- Woody Plant Medium Himedia
- Xylene Chemical SQ 2.5ltr
- Zinc Acid Fast Stains Kit
- Zinc Sulphate SQ 500G Qualigens
- Handwash savlon professional white Can 5L
- Acetic Acid Glacial
- Chlorotex Reagent 100ml (2)
- EDTA N50 Solution 500ml
- Hydrogen Peroxide solution ER 500ml
- Hypersil GOLD C18 Selectivity HPLC Columns
- Lead Acetate Trihydrate
- Mercuric Sulphate 100g
- N Butyl Alcohol ER
- Nitric Acid ER 500ml
- Paraffin Liquid Light
- Potassium Dichromate SQ 500g
- Potassium Iodide ER 250g
- Potassium Nitrate AR 500G
- Sodium Sulphide Hydrate Flakes Iron Free
- Laboratory Instruments
- R-4C Compact Laboratory Centrifuge
- TAB2T Wensar Analytical Balance
- Fluke 59 Max Plus Infrared Thermometer
- R-8M Plus Laboratory Centrifuges
- R-8C PLUS Laboratory Centrifuges
- RWB 6 REMI Water Bath
- Laboratory Silica Crucible
- Analab Automatic Melting Boiling Point Apparatus
- RQT-124A-D Laboratory Stirrers
- RQ-124A-D Laboratory Stirrers
- LMPH12 5 Points pH Meter
- LMPH15 pH Meter With Printer
- TAB1T WENSAR Analytical Balance
- TAB4T Wensar Analytical Balance
- KAB1L Wensar Analytical Balance
- Plastic Blood Lancet
- RQ-130B-D SS Laboratory Stirrers
- RQG-129-D SS Laboratory Stirrers
- Laboratory Instruments
- Ph Orp Electrodes
- Remi Dry Hot Air Oven RD
- RQ-121/D REMI
- Medico Centrifuge R-304
- Medico Centrifuge R-303
- Medico Centrifuge C-854/8
- Medico Centrifuge C-854/4
- Medico Centrifuge C-854/6
- RQ-140/DE Laboratory Stirrers
- COMPACT LABORATORY CENTRIFUGE R 4C
- Laboratory Centrifuges R 8M Plus
- Water Testing SD 90
- Racer Digital Stopwatch, For Laboratory
- ACETEQ DTM-X9 FOLDING FOOD THERMOMETER
- Digital Oven Thermometer
- Digital Refractometer ATAGO Ethylene Glycol Refra
- SD 90 Water Testing
- Rotary Shaker with Brushless Induction Motor
- REMI Dry Hot Air Oven RDHO-50
- Aluminium Alloy 4 In 1 Soil Survey Instrument
- MERA PAL ATAGO Pocket Digital Refractometer
- Hanna Instruments HI 96107 pH meter, 85 G
- Digital Oven Thermometer
- 4 In 1 Soil Survey Instrument
- Aceteq Dtm-X9 Folding Food Thermometer
- Analab Automatic Melting Boiling Point Apparatus
- Analytical Balance KAB1L Wensar
- Analytical Balance KAB4L Wensar
- Analytical Balance TAB1T WENSAR
- Analytical Balance TAB2T Wensar
- Analytical Balance TAB4T Wensar
- Benchtop PH Meter Remi
- Borosil Culture & Petri Dish
- Ceramic Hotplate Magnetic Stirrer
- Compact Laboratory Centrifuge R 4C
- Conductivity Meter
- Digital Refractometer ATAGO Ethylene Glycol Refractometer, PAL-91S, For Industrial
- Digital Refractometer
- Digital Stem Thermometer
- Digital Vernier Caliper
- Fluke 59 Max Plus Infrared Thermometer
- Hanna Instruments HI 96107 pH meter 85 G
- Laboratory Centrifuges Plus R 8C Plus
- Laboratory Centrifuges R 8M Plus
- Laboratory Stirrer Remi
- LMPH10H - pH Meter-3 Points
- LMPH15 - pH Meter-With Printer
- Magnetic Stirring Bar Retriever PTFE
- Measuring Pipette 5ML BOROSIL
- Medico Centrifuge C-852
- Medico Centrifuge C-8544
- Medico Centrifuge C-8546
- Medico Centrifuge C-8548
- Medico Centrifuge R-303
- Medico Centrifuge R-304
- MERA PAL ATAGO Pocket Digital Refractometer
- Microlit E-BURETTE With 3 Calibrated Pre Set Speeds
- Microplate Shaker MPS 1
- Mini Rotary Shaker RS 12R
- Mini Rotary Shaker RS-18 Plus
- Moisture Analyzer Remi
- Petri Dish 100x17MM BOROSIL
- Polylab 55215 Vacuum Desiccator
- Racer Digital Stopwatch, For Laboratory
- Remi Dry Hot Air Oven RDHO 80
- REMI Dry Hot Air Oven RDHO-50
- Remi Rs 12 Plus Mini Rotary Shaker
- REMI Water Bath RWB 6
- Rotary Shaker with Brushless Induction Motor
- RQ 128 D Laboratory Stirrers
- RQ-121D REMI
- RQ-122D Laboratory Stirrers
- RQ-124AD Laboratory Stirrers
- RQ-126D Laboratory Stirrers
- RQ-127AD Laboratory Stirrers
- RQ-127D Laboratory Stirrers
- RQ-128BD Laboratory Stirrers
- RQ-128DD Laboratory Stirrers
- RQ-129D Laboratory Stirrers
- RQ-130BD Laboratory Stirrers
- RQ-134HD Laboratory Stirrers
- RQ-134LD Laboratory Stirrers
- RQ-134SD Laboratory Stirrers
- RQ-140D FP Laboratory Stirrers
- RQ-140DE Laboratory Stirrers
- RQG-121D Laboratory Stirrers
- RQG-126D Laboratory Stirrers
- RQG-128AD Laboratory Stirrers
- RQG-129D Laboratory Stirrers
- RQM-122R REMI
- RQT-124AD Laboratory Stirrers
- RQT-127A-D Laboratory Stirrer
- RQT-127D Laboratory Stirrers
- RQT-134HD Laboratory Stirrers
- RS 24 Plus Rotary Shaker
- RSB-12 Water Bath Shakers
- Spectrophotometer Remi
- Tarsons 20c Pcr Mini Cooler
- Thumb Press Dispensing Dropper Pasteur Pipettes
- Total Hardness Test Kit (250 Test)
- Tube Roller Mixer SeroRoll 6 REMI
- Turbidity Meter Remi
- UV-VIS Spectrophotometer Labman
- VWR TLC Aluminium Plates
- Himedia Vails Laboratory Chemicals
- Hydranal Water Standard 0.1 Honeywell
- Co Trimoxazole Sulpha Trimethoprim Himedia
- M065 Agar Deoxycholate Citrate
- M377 Agar Lysine Iron
- Nutrient Agar
- M001 Agar Nutrient
- Nutrient Agar
- M118 Mannitol Salt Agar Base
- CLED Blue Agar Bromo Thymol
- M173 500g Mueller Hinton Agar Powder
- M073 500g Blood Agar Base
- M099 100g Simmons Citrate Agar
- M962 R2A Agar
- M049-500G Violet Red Bile Agar
- M009-500G Fluid Thioglycollate
- M008 500G Macconkey Agar Powder
- M173 500g Mueller Hinton Agar
- M002 500G Nutrient Broth Powder
- MH081 Sabouraud Dextrose Agar Powder
- GRM026 Agar Bacteriological Grade Powder
- GRM199 500g Hi-A Glycine
- REINFORCED CLOSTRIBIAL AGAR
- CDTA monohydrate Hi AR ACS
- Plate Count Agar Standard Methods Agar
- Dey Engley Neutralizing Broth
- Yeast Glucose Agar
- Violet Red Bile Agar
- HiCrome Candida Differential Agar
- EMB Agar
- Kovacs Indole Reagent
- Gordon McLeod Reagent (Oxidase reagent)
- Peptone, Bacteriological
- Optochin 5 mcg 50 discs
- SD037-1VL Tetracycline
- SD065-1VL Ceftriaxone
- SD278-1VL Tigecycline
- SD216 Levofloxacin
- SD051-1VL Clindamycin
- SD211 Cefixime
- SD013-1VL Erythromycin
- SD082-1VL Amikacin
- SD215-1VL Linezolid
- SD003-1VL Bacitracin
- SD023-1VL Nitrofurantoin
- SD060-1VL Ciprofloxacin
- Cefoxitin (Cephoxitin)
- SD219-1VL Cefepime
- SD062-1VL Ceftazidime
- SD057-1VL Norfloxacin
- SD727-1VL Meropenem
- Violet Red Bile Agar
- GN Broth Medium 11
- Anaerogas Pack 3.5L
- Reinforced Clostridial Agar
- HiCrome Candida Differential Agar
- Anaerogas Pack 3.5L
- Nichrome Loop D-2
- Sabouraud Dextrose Broth, MH033
- Casein Soyabean Digest Agar
- Peptone Bacteriological
- HiCrome Chromogenic Coliform
- Simmons Citrate Agar
- Simmons Citrate Agar
- EMB Agar
- MacConkey Agar
- Listeria Selective Enrichment Broth
- Dey Engley Neutralizing Agar
- Mannitol Salt Agar
- Buffered Sodium Chloride
- Xylose Lysine Deoxycholate Agar XLD Agar
- MacConkey Broth
- MacConkey Broth Double
- Simmons Citrate Agar
- Simmons Citrate Agar
- Soyabean Casein Digest Agar 500G
- Amoxicillin .
- Potassium tellurite Hi LR
- AK Agar No 2 (Sporulating Agar) (Arret and Kirshbaum Medium)
- Modified Phosphate Buffer
- Fluid Casein Digest Soya Lecithin Medium (Twin Pack)
- Antibiotic Assay Medium No 35
- Antibiotic Assay Medium No 32
- Nichrome Loop-D-2
- Simmons Citrate Agar
- Antibiotic Assay Medium No 19
- Antibiotic Assay Medium No 3 Assay Broth
- MP 5 Medium
- Sabouraud Chloramphenicol Agar
- Agar powder Bacteriological grade
- Medium 8 MacConkey Agar
- Sabouraud Chloramphenicol Agar
- Antibiotic Assay Medium No 3 Assay Broth
- MP 5 Medium , Packaging Details: Plastic Bottle
- Antibiotic Assay Medium No 19 , Packaging Details
- Modified Phosphate Buffer , Packaging Details: Bot
- Antibiotic Assay Medium No 35 , Packaging Details
- Antibiotic Assay Medium No 9 Polymyxin Base Agar , Packaging Details: Bottle
- AK Agar No.2 (Sporulating Agar) (Arret and Kirshba
- Fluid Casein Digest Soya Lecithin Medium (Twin Pack) , Packaging Details: Plastic Bottle
- Antibiotic Assay Medium No 36
- Vogel Johnson Agar Medium
- Antibiotic Assay Medium No 1 Seed Agar
- Antibiotic Assay Medium No 2 Base Agar
- Soybean-Casein Digest Agar
- Antibiotic Assay Medium No 11
- Antibiotic Assay Medium No 32
- Amoxyclav AMC 1VL
- Ammonium persulphate Hi AR ACS
- Potassium persulphate Hi AR ACS
- Ammonium persulphate Hi AR ACS , Packaging Details: Plastic Bottle
- Coagulase Plasma 0.1gm per vial , Packaging Details: Box
- Pseudomonas Agar For Fluorescein , Packaging De
- GN Broth Medium 11
- Simmons Citrate Agar 100g Himedia M099
- Eosin yellow water soluble Hi Cert ACS
- Disposable Forceps 12.5CM
- Cetrimide Agar, 100G
- Steam Indicator Tape
- Chloramphenicol Yeast Glucose Agar
- Gentamicin 10mcg VL Himedia
- Cefotaxime Cephotaxime 30mcg Himedia
- Imipenem 10mcg Himedia
- Ampicillin 10mcg Himedia
- Vancomycin 30mcg Himedia
- Tigecycline .
- Piperacillin Tazobactam
- HiCrome Chromogenic Coliform agar
- trans Diaminocyclohexan tetraacetic acid monohy
- Stainless Steel Forceps, Blunt Himedia
- Norfloxacin 10mcg 1VL Himedia
- Tetracycline 30 mcg 1VL Himedia
- HEXAMINE (methenamine, hexamethylene tetrami
- Filtration Funnel Polypropylene w GCN Me
- Oxidase Discs (50 discs / vl)
- Sterile Cotton Swab
- Sterile Disposable Petri Plates
- Gram Stains Kit K001
- HiCrome Chromogenic Coliform agar
- Violet Red Bile Glucose Agar
- Triple Sugar Iron Agar
- Cedarwood Oil, For Microscopy
- Columbia Agar,500G
- Reinforced Medium for Clostridia
- Rappaport Vassiliadis Salmonella Enrichment Bro
- Diagnostic Stuart s Urea Broth Base
- Sterile, Disposable L Spreader
- R-2A Agar
- Cetrimide Agar 500G
- Blood Agar Base Himedia
- Agar Agar
- Lysine Iron Agar HIMEDIA
- Mannitol Salt Agar Base HIMEDIA
- Antibiotic Assay Medium No 36 , Packaging Details
- Macconkey Agar Himedia , Packaging Details: Bottle
- Oxidase Discs (50 discs / vl)
- Eosin yellow water soluble Hi Cert ACS
- Potassium Sulphate
- Plate Count Agar (Standard Methods Agar) ,
- Fluid Thioglycollate Medium Thioglycollate Medium
- HiCrome Chromogenic Coliform agar (CCA Agar) ,
- Soyabean HiVe Medium, Sterile Powder (Tryptone
- Harris Haematoxylin , Packaging Details: 125ML
- Kovacs Indole Reagent , Analytical Grade
- Peptone, Bacteriological , Packaging Details: Bottle
- Agar Medium S (R2A Agar) , Packaging Size: 500 g
- Buffered Sodium Chloride Peptone Solution, , 98%
- Chemical Grade Powder Nutrient HiVeg Agar , 500
- Cefuroxime CXM 30 mcg
- Chloramphenicol 30mcg 1VL Himedia ,
- Ofloxacin OF 5 mcg
- Teicoplanin TEI 30 mcg
- Colistin (Methane Sulphonate) CL 10 mcg
- Tetracycline TE 30 mcg
- Amphotericin B 32mcg
- Amikacin AK 30 mcg
- Fluconazole FLC 256mcg
- Tobramycin TOB 10 mcg
- Colistin (Methane Sulphonate) CL 10 mcg
- Amoxyclav (Amoxicillin Clavulanic acid) AMC 30 mcg
- Caspofungin CAS 32mcg
- Lithium carbonate Hi AR ACS
- Itraconazole ITR 32mcg
- Antibiotic Assay Medium No 40
- Soyabean Casein Digest Medium (Tryptone Soya Br
- Lithium carbonate Hi AR ACS
- Aztreonam AT 30 mcg
- Amoxyclav (Amoxicillin Clavulanic acid) AMC 30 m
- Azithromycin 15mcg 1VL
- Colistin (Methane Sulphonate) CL 10 mcg
- Nitrofurantoin 300mcg 1VL Himedia
- Colistin (Methane Sulphonate) CL 10 mcg
- Ofloxacin OF 5 mcg
- Laboratory Consumables
- Non Absorbent Cotton Roll
- Parafilm M Roll
- Kimtech Purple Nitrile Exam Gloves
- Parafilm Grafting Tape
- Nddb Milk Adulteration Test Kit
- DELMOS Milk Adulteration Kit
- 521010 2-200ul Tarsons Micro Tips
- 250ML Laboratory Heating Mantle
- 100ML Ferroin Indicator Solution
- Aluminium Fine Powder
- 10ul Micropipette Tips
- 200ul Micropipette Filter Tips
- Petri dishes with triple vent Radiation Sterile
- 80 mm Plate Glass Petri Dish
- Glass Petri Dish
- Polyethylene Disposable Hand Gloves
- Blue Disposable Bouffant Cap
- Three Ply Non Woven Disposable Face Masks
- Mce Membrane Filter 0.45 Um Pore Size
- Bowie Dick Test Pack
- Laboratory Heating Mantle, For Air Conditioners, Stainless Steel
- Laboratory Silica Crucible 100ML
- Supertek Cellulose Nitrate Membrane Filter Dia 47 m
- Whatman 1822-037 Grade GF/C Glass Microfiber Filt
- Dpd No 4 Tablets
- LABORATORY TRAY
- Laboratory Burette Stand
- Parafilm M Roll
- Round Plastic 7 Inch Test Tube Washing Brush, Fo
- Spatula Ended Spoon
- Steel Dissecting Forceps Orthopedic Instrument
- Semi Automatic SS Spirit Lamp
- A mortar & pestle 4 Inch
- SRL White To Off White Dish Evaporating Porcelain, 100ml, OD 75mm, RB, Deep with Spout , For Chemical Laboratory
- Laboratory Silica Crucible 50ML
- Pipette Rack Horizontal, PP Tarsons
- TARSONS SPINWIN Tube Conical
- Laboratory Plastic Beakers
- Plastic Embedding Cassette
- Digital Stem Thermometer
- Parafilm Grafting Tape, 2 Inch X 225 Feets
- Reagent Bottle Wide Mouth 500ML
- Reagent Bottle Wide Mouth 1000ML
- Reagent Bottle Wide Mouth 250ML
- Reagent Bottle Wide Mouth 125ML
- Plastic Measuring Cylinder
- Measuring Cylinder Class B, PP 355040
- Narrow Mouth Wash Bottle LDPE with PP closure a
- Milk Adulteration Kit DELMOS
- Industrial Grade Sulphuric Acid SQ 5L , For Cleaning Agent, 98%
- Glass White Silica Crucible Capacity 15ml
- BD BBL IsoVitaleX Enrichment
- Spinwin Tube Conical Bottom 500041 TARS
- Non-Sterilised Centrifuge Tube Conical
- Glass White Silica Crucible Capacity 15ml ,
- 10ul Micropipette Filter Tips
- 1000ul Blue Micropipette Tip
- Spatula 8" (Pack of 6)
- Wash Bottle, LDPE TARSONS
- Wide Mouth Bottle, HDPE with P
- 18x18 mm Microscopic Cover Glass
- Tarsons Micro Tips 521010 2-200ul
- Beaker, PP 10000ML
- Turmeric Indicator Papers
- A mortar & pestle 4 Inch
- A mortar & pestle 4 Inch
- TEST TUBE STAND
- Wash Bottle LDPE TARSONS
- Glass Van Sterile Surgical Blad
- Wide Mouth Bottle, PP with PP closure
- Micro Tips Bulk
- Wide Mouth Bottle, LDPE with PP clo
- 10ul Micropipette Filter Tips Box
- Non Absorbent Cotton Roll
- RingBio Aflatoxin M1 Rapid Strip Test Kit
- Wide Mouth Bottle, LDPE with PP closure
- Tarsons Measuring Beaker ,
- Whatman qualitative filter paper Grade 4
- TEST TUBE STAND (3 TIER) P.P
- Steel Dissecting Forceps Orthopedic Instrument
- 10ul Micropipette Filter Tips Box
- 200ul Micropipette Filter Tips (2)
- 200ul Micropipette Filter Tips
- A Mortar & Pestle 4 Inch
- ALUMINIUM FINE POWDER 98% Extra Pure Loba
- Ammonia solution Merck
- Anaerogas Pack 3.5L
- Bacillocid Special 500ml
- Blue Disposable Bouffant Cap
- Borosil 15ml Plain Centrifuge Tube
- Borosil S Line Petri Dish (2)
- Borosil S Line Petri Dish
- Bromocresol Green Indicator Powder 5g
- Centrifuge Tube 50 Ml
- Delmos Milk Adulterant Strips kit (2)
- Delmos Milk Adulterant Strips kit
- Disposable Buffent Cap
- Ferroin Indicator Solution ER 100ML Qualigens
- Filtration Funnel Polypropylene w GCN Membrane
- Glass Van Sterile Surgical Blade
- Glass White Silica Crucible Capacity 15ml
- Hypersil GOLD C8 HPLC Columns
- John Okay Water Proof Paper Emery paper
- Kimtech Purple Nitrile Exam Gloves
- Indicator Papers Full Range Qualigens
- Labolene Neutral 500ml
- Laboratory Heating Mantle 250ml
- Laboratory Heating Mantle
- Laboratory Parafilm Tape
- Laboratory Spirit Lamp
- Laboratory Tray Polylab
- Lovibond Vario Reagents
- Micro Tips Bulk (2)
- Micro Tips Bulk 521026
- Micro Tips Bulk
- Micropipette Tips 10ul
- Microscope Cover Glass
- Milk Adulteration Kit Delmos 100 Strip set
- Milk Adulteration Kit DELMOS
- Narrow Mouth Wash Bottle LDPE with PP closure and draw tube
- Nddb Milk Adulteration Test Kit Large Size
- Non Absorbent Cotton Roll
- Non Absorbent Cotton Wool 500g
- Nutrient Agar Himedia
- Nylon Test Tube Brush (2)
- Nylon Test Tube Brush
- Parafilm M Roll
- Petri dishes with triple vent Radiation Sterile (2)
- Petri dishes with triple vent Radiation Sterile
- Ph Orp Electrodes
- Pipette Measuring 2 ml Borosil
- Plastic Embedding Cassette
- Polyethylene Disposable Hand Gloves
- Randisc Nylon Syringe Filter For Laboratory
- RingBio Aflatoxin M1 Rapid Strip Test Kit
- Rubber Pipette Bulbs For Chemical Laboratory
- Sartorius YDP50 Compact Dot Matrix Printer
- Semi Automatic SS Spirit Lamp
- Sorbitol Test Solution
- Spatula 10 Pack
- Spatula Ended Spoon 10CM
- Stainless Steel Forceps Blunt Himedia
- Sterile Cotton Swab Himedia
- Sterile Cotton Swab
- Sterind Bowie And Dick Test Pack 121 Deg 15 Mins
- Tarsons 200 1000 ul Polypropylene Micro Tip
- TDS 442 Tandard Solutions 50ppm (2)
- TDS 442 Tandard Solutions 50ppm
- TEST TUBE STAND
- Test tube with rim Borosil (2)
- Test Tube with Rim BOROSIL
- Tips For Micropipette Micro Tips 2-200 ul 521010
- TLC Plates Silica Gel 60 F254 Aluminum
- Whatman Grade 40 Ashless Quantitative Filter Paper
- Wide Mouth Bottle HDPE with PP closure
- Wide Mouth Bottle LDPE with PP closure (2)
- Wide Mouth Bottle LDPE With PP Closure
- Wide Mouth Bottle PP with PP closure
- Sigma Aldrich Chemical
- LISPHER100 RP18E 5MYM CART250-4
- 100GM 1332-58-7 Halloysite nanoclay
- 100GM 57-09-0 Hexadecyltrimethylammonium Bromide
- 78-10-4 Tetraethyl Orthosilicate Reagent
- 513-35-9 2-Methyl-2-butene
- 3452-97-9 3 5 5 Trimethyl 1 Hexanol
- 544-76-3 N Hexadecane
- 7699-45-8 Zinc Bromide
- 60-12-8 2-Phenylethanol
- 79-07-2 2 Chloroacetamide
- 7758-95-4 Lead II Chloride
- 61-82-5 3-Amino-1H-1,2,4-triazole
- 78-10-4 Tetraethyl Orthosilicate
- 83-46-5 Beta Sitosterol
- 60-24-2 2 Mercaptoethanol
- 2 Amino 2 Methyl 1 Propanol , Analytical AR Grade
- Benzene ACS Reagent
- Sodium metasilicate pentahydrate 250G
- Universal indicator solution pH 4-11
- Rhodamine B
- Hexadecane , Packaging Details:
- Hexadecyltrimethylammonium bromide 100GM
- Laboratory Glasswares
- Borosil Low Form Beaker With Spout 500ml
- 500ml Beaker Low Form With Spout
- Erlenmeyer Conical Flask
- 8852 Pear Shape Splash Head Sloping
- Laboratory Plastic Beaker
- Narrow Mouth Reagent Bottle With Screw Cap
- Solution Bottle With Tooled Neck
- 250ml Glass Bottle
- 8 Inch Spatula
- Polylab 55215 Vacuum Desiccator
- 10ml Measuring Cylinder
- Borosil Test Tube Without Rim
- Drum Sampling Pipette
- Oxygen Combustion Flask
- LOW FORM BEAKER With Spout 500ML
- ERLENMEYER CONICAL FLASK 250ML Narrow Mouth, With Rim , For Chemical Laboratory, 250 ml
- SEPARATING FUNNEL PEAR SHAPE With Boroflo S 1000ML
- SEPARATING FUNNEL PEAR SHAPE With Boroflo S
- LOW FORM BEAKER 250ML With Spout BEAKER With Sp
- LOW FORM BEAKER With Spout ,500ML
- LOW FORM BEAKER With Spout,150ML
- Test Tube Without Rim,16*50
- ERLENMEYER CONICAL FLASK,250ML Narrow Mouth, With Rim , For Chemical Laboratory, 250 ml
- LOW FORM BEAKER With Spout
- LOW FORM BEAKER With Spout, 100ML M BEAKER With Spout, 100 ml
- LOW FORM BEAKER With Spout 250ML
- LOW FORM BEAKER With Spout M BEAKER With
- LOW FORM BEAKER With Spout, 50ML
- VOLUMETRIC FLASKS Class A,25ML Narrow Mouth, Clea
- VOLUMETRIC FLASKS Class A, 1000ML Narrow Mouth, Clear, With Individual Calibration Certificate
- CONDENSER FOR EXTRACTION APPARATUS
- REAGENT BOTTLES 100ML Narrow Mouth With Scre
- REAGENT BOTTLES 250ML Narrow Mouth With Scre
- VOLUMETRIC FLASKS Class A,100ML Narrow Mouth, Clear, with Individual Calibration Certificate
- VOLUMETRIC FLASKS Class A,500ML Narrow Mouth, Cl
- measuring cylinder 10ml (riviera)
- Test Tube Borosilicate Glass
- Test Tube Borosilicate Glass
- SOLUTION BOTTLES With Tooled Neck
- Test Tube With Rim
- TEST TUBES Without Rim 5ML
- ROUND BOTTOM FLASK Narrow Mouth, Short Neck With I/C Joint
- measuring cylinder 10ml (riviera)
- Nessler Cylinder
- MOHR PIPETTES Class A
- Test Tube Without Rim
- Test Tube With Rim
- VOLUMETRIC FLASKS Class A, Narrow Mouth, Clear, with Individual Calibration Certificate
- CYLINDERS Class A, Hexagonal base, Pour out, wit
- CYLINDERS Class A, Hexagonal base, Pour out, with Individual Calibration Certificate
- WEIGHING BOTTLES With I/C Glass Lid
- IODINE FLASK With I/C Glass Stopper
- VOLUMETRIC FLASKS Class A, Narrow Mouth, Clear, with Individual Calibration Certificate
- CULTURE TUBE Flat Bottom, Clear, With PP Cap
- ERLENMEYER (CONICAL) FLASK Narrow Mouth, With Rim
- VOLUMETRIC FLASKS Class A, Narrow Mouth, Clear, with Individual Calibration Certificate
- NESSLER CYLINDERS
- MOHR PIPETTES Class B, White Marking
- SEPARATING FUNNEL PEAR SHAPE With Boroflo S
- WEIGHING BOTTLES With I/C Glass Lid
- CYLINDERS Class A, Hexagonal base, Pour out, with Individual Calibration Certificate
- REAGENT BOTTLES Narrow Mouth With Screw Cap, Amber
- VOLUMETRIC FLASKS Class A, Narrow Mouth, Clear, with Individual Calibration Certificate
- VOLUMETRIC FLASKS Class A, Narrow Mouth, Clear, with Individual Calibration Certificate
- FLAT BOTTOM EVAPORATING DISHES With Spout
- REAGENT BOTTLES Narrow Mouth With I/C Glass Stopper, Amber
- IODINE FLASK With I/C Glass Stopper
- REAGENT BOTTLES Narrow Mouth With I/C Glass S
- Borosil Test Tube Without Rim 12 X75 5
- LOW FORM BEAKER With Spout M BEAKER With
- ERLENMEYER CONICAL FLASK Narrow Mouth, With Rim , For Chemical Laboratory, 250 ml
- REAGENT BOTTLES Narrow Mouth With Screw Cap
- DEAN STARK DISTILLING APPARATUS 3602005
- Bottle Mojonnier Glass Extraction Flask ,
- Borosil S Line Petri Dish
- Silica Gel 60 F254 25 TLC Plates 20 x 20 20 X 20
- REAGENT BOTTLES Narrow Mouth With Screw Cap, , 100 ml
- REAGENT BOTTLES Narrow Mouth With Sc
- Diphenylamine 4 Sulfonic Acid Barium Salt
- SOLUTION BOTTLES With Tooled Neck
- DEAN STARK DISTILLING APPARATUS
- DEAN STARK DISTILLING APPARATUS
- DEAN STARK DISTILLING APPARATUS
- ROUND BOTTOM FLASK Narrow
- VOLUMETRIC FLASKS Class A Narrow Mouth, Cle
- REAGENT BOTTLES Narrow Mouth
- REAGENT BOTTLES Narrow Mouth With Screw Ca
- SOLUTION BOTTLES With Tooled Neck Amber
- BURETTES 10ML
- Beaker Low Form With Spout 500ml
- Silica Crucible Infusil 50ML
- Lab Watch Glass
- CYLINDERS Class A Hexagonal Base with I/
- Glass Petri Dish
- CULTURE TUBE Flat Bottom, Clear, With PP Cap , 15 ml
- IODINE FLASK With I/C Glass Stopper
- ERLENMEYER (CONICAL) FLASK
- FLAT BOTTOM EVAPORATIN
- MOHR PIPETTES Class A
- MOHR PIPETTES Class B, White Marking
- WEIGHING BOTTLES With IC Glass Lid
- Drum Sampling Pipette
- BURETTES With Boroflo stopcock Class B
- WATCH GLASSES S - Line BOROSIL
- Glass DEAN STARK DISTILLING APPARATUS , Fo
- Glass Butyro Meter For Milk Testing At 65 De
- Glass Pipette Milk 10.75 ISI Mark Lmmco, 15 Gm
- 150 mm Test Tube With Rim , 25 ml
- Nessler Cylinders 50 Ml
- DEAN STARK DISTILLING APPARATUS (2)
- Beaker Low Form With Spout 500ml
- Borosil Beaker Low Form With Spout
- Borosil Bottle Reagent Screw Cap GR
- Borosil Culture Tube Flat Bottom Clear With PP Cap 10ml
- Borosil Culture Tube Flat Bottom Clear With Pp Cap
- Borosil Glass Bottle
- Borosil Glass Petri Dish
- Borosil Glass Reagent Bottle250ml
- Borosil Glass Reagent Bottles 500ml
- Borosil Glass Stirrer Rod (2)
- Borosil Glass Stirrer Rod
- Borosil Iodine Flask With Ic Glass Stopper (2)
- Borosil Iodine Flask With IC Glass Stopper
- Borosil Lab Watch Glass
- Borosil Low Form Beaker With Spout (2)
- Borosil Low Form Beaker With Spout (3)
- Borosil Low Form Beaker with Spout 150ml
- Borosil Low Form Beaker With Spout
- Borosil Measuring Cylinder
- Borosil Round Bottom Flask Narrow Mouth
- Borosil Solution Bottles With Tooled Neck Amber
- Borosil Splash Head Sloping Pear Shape
- Borosil Test Tube Borosilicate Glass
- Borosil Test Tube With Rim 12 X 100mm 7ml
- Borosil Test Tube With Rim
- Borosil Test Tube Without Rim 12 X75 5ml
- Borosil Test Tubes With Rim
- Borosil Test Tubes Without Rim 5ml
- Borosil Volumetric Flasks Class A Narrow Mouth Clear
- Borosil Weighing Bottles With IC Glass Lid (2)
- Borosil Weighing Bottles With IC Glass Lid
- Borosilicate Glass Beaker 50ml
- Borosilicate Reagent Bottles Ambar Screw Cap Capacity 25ml
- Borosilicate Reagent Bottles Ambar Screw Cap Capacity 100ml
- Borosilicate Reagent Bottles Ambar Screw Cap Capacity 250ml
- Borosilicate Reagent Bottles Ambar Screw Cap Capacity 500ml
- Bottle Mojonnier Glass Extraction Flask
- Burette Rotaflow SCock Cl-B
- Burettes With Boroflo Stopcock
- Buretts 10ml
- Condenser For Extraction Apparatus
- Conical Flask Borosilicate Glass For Laboratory Size 250 ML
- Cylinders Class A Hexagonal base Pour out with Individual Calibration Certificate (2)
- Cylinders Class A Hexagonal Base Pour out with Individual Calibration Certificate
- Cylinders Class A Hexagonal Base With IC Stopper Borosil
- Dean Stark Distilling Apparatus
- Drum Sampling Pipette
- Erlenmeyer Conical Flask Narrow Mouth With Rim (2)
- Erlenmeyer Conical Flask Narrow Mouth With Rim
- Glass Pipette Milk 10.75 ISI Mark Lmmco, 15 Gm
- Glass Reagent Bottle Capacity 250ml
- Hypersil BDS C18 Columns
- Laboratory Silica Crucible 50ml
- Laboratory Silica Crucible 100ml
- Laboratory Silica Crucible,
- Measuring Cylinder 10ml Riviera
- Measuring Cylinder Class B PP
- Mohr Pipettes Class A
- Oxygen Combustion Flask As Per Usp With 99.9% Platinum Basket
- Oxygen Combustion Flask Platinum Basket
- Pipette Rack Horizontal, PP Tarsons
- Plastic Measuring Cylinder
- Reagent Bottle Narrow Mouth With Screw Cap 150ml
- Reagent Bottles Narrow Mouth With IC Glass S
- Reagent Bottles Narrow Mouth With IC Glass Stopper Amber (2)
- Reagent Bottles Narrow Mouth With IC Glass Stopper Amber (3)
- Reagent Bottles Narrow Mouth With Ic Glass Stopper Amber Borosil (2)
- Reagent Bottles Narrow Mouth With Ic Glass Stopper Amber Borosil
- Reagent Bottles Narrow Mouth With IC Glass Stopper Amber
- Reagent Bottles Narrow Mouth With Screw Cap Amber (2)
- Reagent Bottles Narrow Mouth With Screw Cap Amber Borosil (2)
- Reagent Bottles Narrow Mouth With Screw Cap Amber Borosil
- Reagent Bottles Narrow Mouth With Screw Cap Amber
- Reagent Bottles Narrow Mouth With Screw Cap
- Round Bottom Flask Narrow Mouth Short Neck With IC Joint
- Silica Crucible Infusil 50ml
- Solution Bottles With Tooled Neck
- Spatula 8 Pack of 6
- Splash Head Sloping Pear Shape (2)
- Splash Head Sloping Pear Shape
- Splash Head Sloping Pear Shape7
- Tarsons Micro Tips 200-1000 ul 521020
- Test Tube With Rim
- Test Tube Without Rim Borosil
- Volumetric Flasks Class A Narrow Mouth Clear with Individual Calibration Certificate (2)
- Volumetric Flasks Class A Narrow Mouth Clear with Individual Calibration Certificate (3)
- Volumetric Flasks Class A Narrow Mouth Clear With Individual Calibration Certificate
- Watch Glasses S-line Borosil
- MERCK
- Hydrochloric Acid Lr
- Phosphorus Standard for ICP"
- Paraffin Liquid Light
- Crystal Silver Nitrate 100gm (MERCK)
- CombiTitrant 5
- Immersion oil acc to DIN 58884 for microscopy
- Combi Titrant
- Oil Immersion
- Ammonium heptamolybdate tetrahydrate
- Silver Nitrate 100gm MERCK
- Ammonium Iron (II) Sulfate Hexahydrate
- Sulphuric Acid 98% Emplura
- Tetrachloroethylene for spectroscopy Uvasol
- Potassium Dichromate EMPLURA
- Sodium azide EMPLURA
- Ammonia buffer solution
- Merck Isopropyl Alcohol (IPA)
- Potassium iodide for analysis EMPARTA
- Buffer solution (potassium hydrogen phthalate), tra
- Chloroform EMPLURA MERCK 500ML
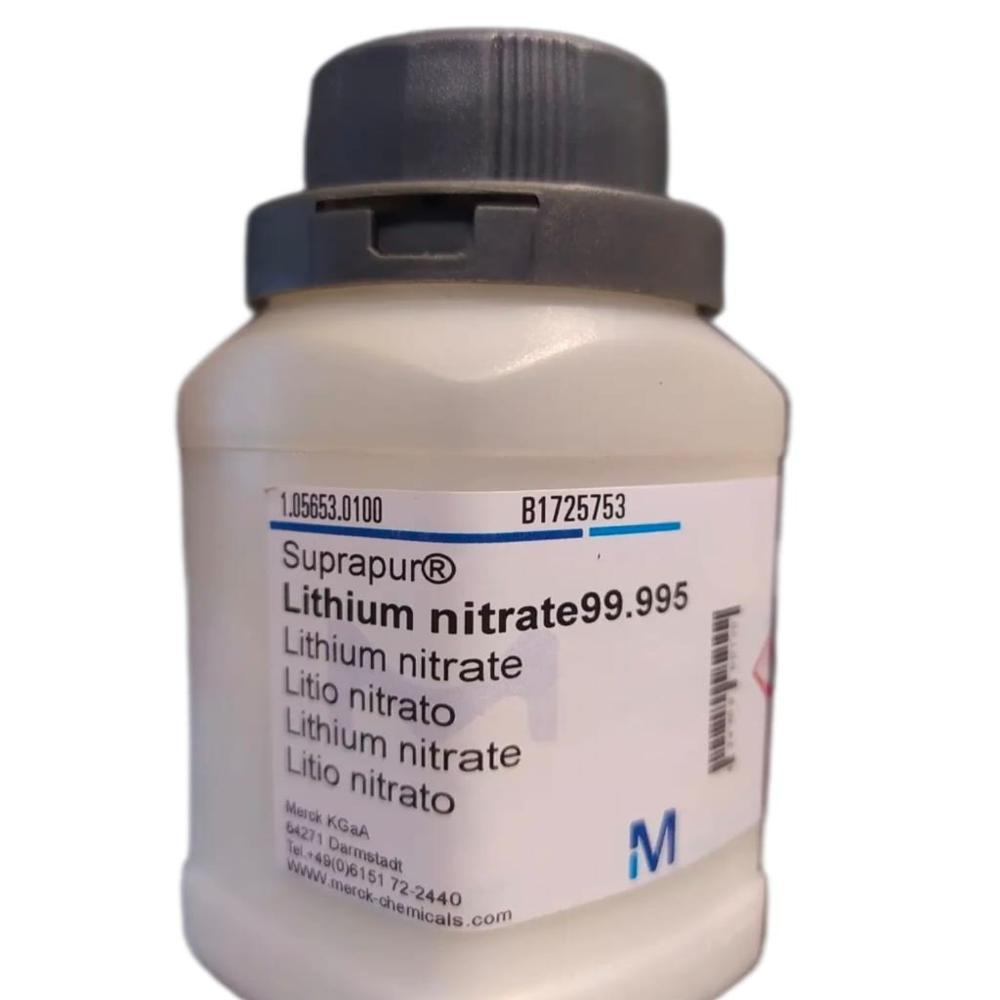
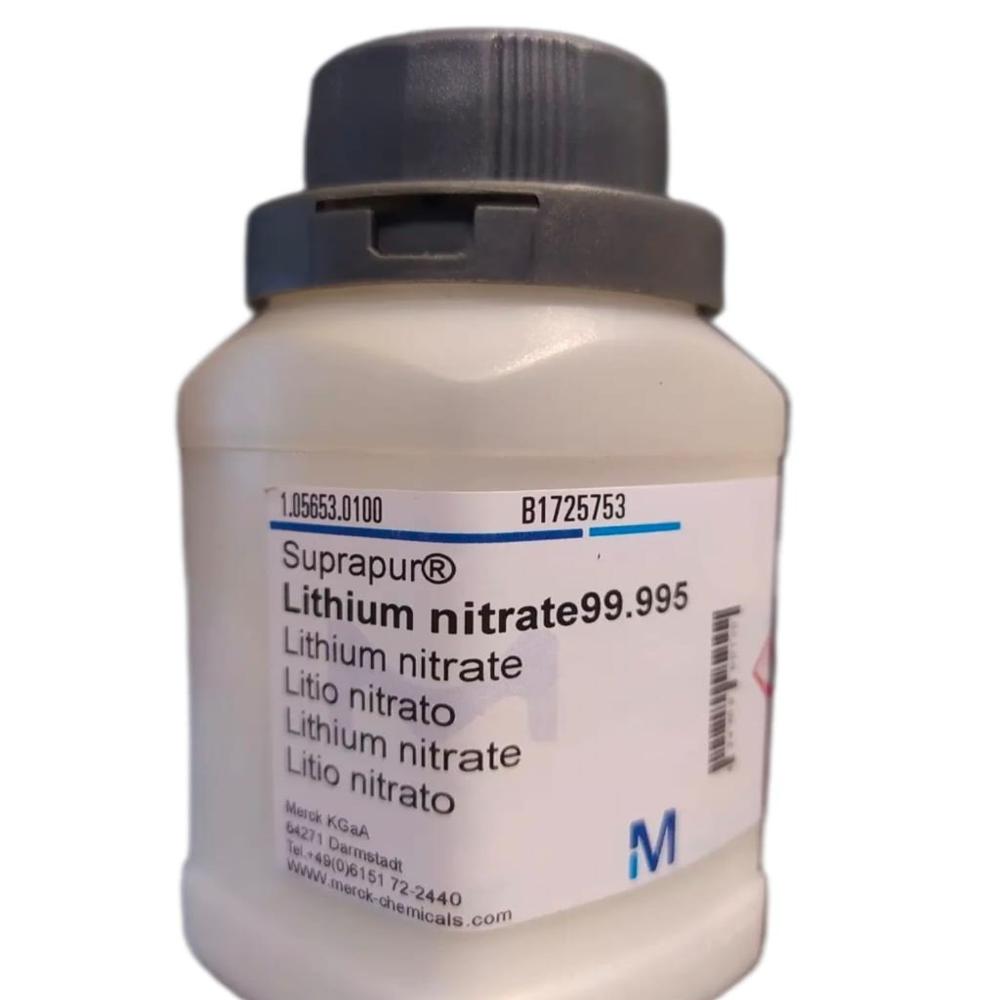
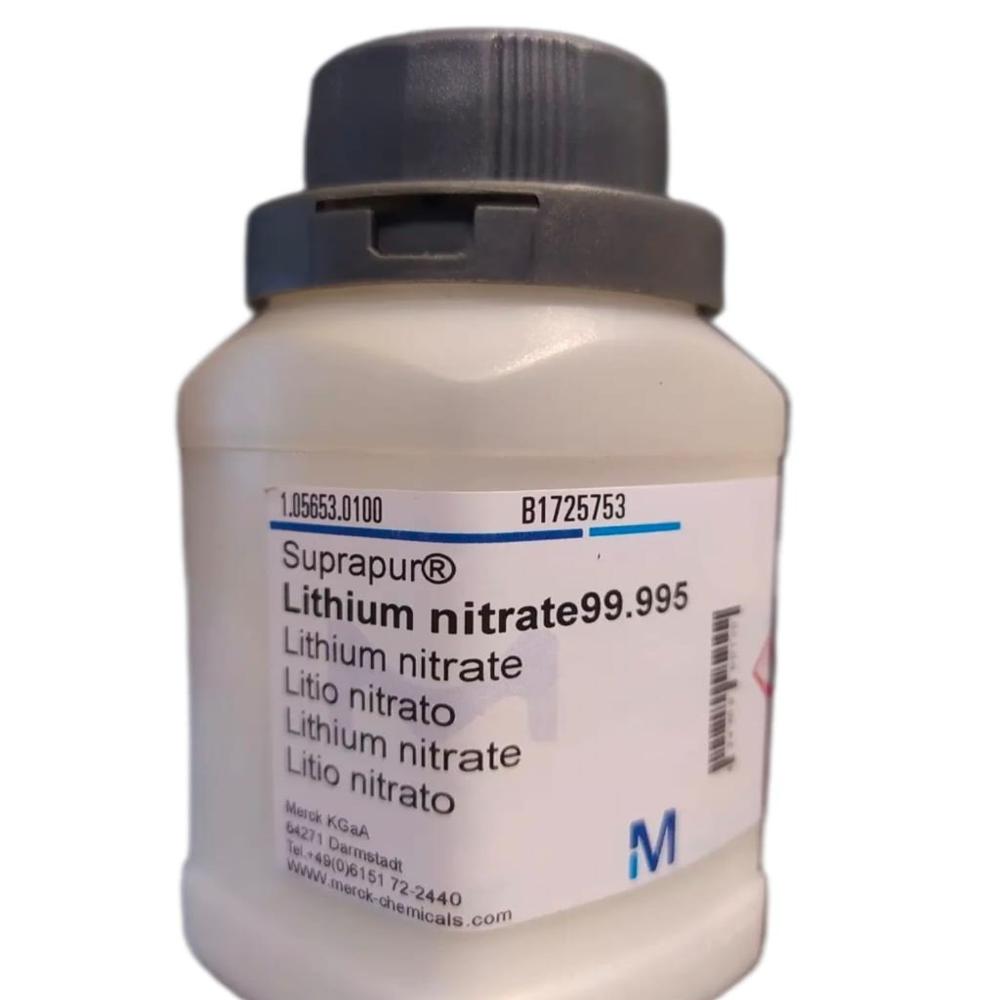
- Lithium Nitrate 99.995 Suprapur
- N-HEXANE EMPLURA 2,5 L
- Potassium Chloride Solution 3 Mol/L
- Buffer solution Certipur
- Methanol for Chromatography Gradient Grade MER
- Karl Fischer s solution pyridine free single solution
- Boron trifluoride methanol complex for synthesis
- Sodium Tartrate Dihydrate
- Extra Pure Gycerol
- MILLIPORE DURAPORE PVDF 45UM WH PL
- Acetonitrile HPLC Grade
- Supelclean PSA SPE Bulk Packing 100G
- Powder Ammonium heptamolybdate tetrahydrate E
- Merck Acetone EMPLURA 25L Lr Grade
- Silver nitrate for analysis EMPARTA ACS RTA ACS, I
- Mce Membrane Filter 0.45 Um Pore Size
- EMPLURA Tin Metal
- Nitric Acid EMPARTA
- Merck Potassium Iodide Emparta Merck
- THERMO FISHER
- Water Testing Chemical
- Milipore Membrane Filter
- HVLP04700 Durapure PVDF.45UM WH PL 47MM
- PVDF Membrane Filter 0.22 um Pore Size Millipore
- Blue Membrane Filters Pore Size 045 Micron Diameter 47 Mm
- MCE Membrane Filter 5.0 um Pore Size
- MCE Membrane Filter 1.2 Um Pore Size Millipore
- Millipore Filter Papers
- MCE Membrane Filter 0.22 um Pore Size
- SMWP04700 MILLIPORE Filter paper
- Supertek Cellulose Nitrate Membrane Filter Dia 47 mm, Pore Size 0.45 um, MCN04-N47 (Pack of 100)
- MF Millipore Membrane Filter 5 um pore size SMWP04700 Diameter 47mm
- PVDF Membrane Filter 0.45 um Pore Size HVLP04700
- HAWP04700 Membrane Filter 0.45m 47mm Diamter
- Silver Nitrate
- Silver Nitrate Merck
- Silver Sulphate Ar Loba
- Silver Nitrate Acs 25g
- Silver Nitrate 25g
- Silver Nitrate ER 25g
- Silver Nitrate N-10 C V S Qualigens
- Silver Nitrate 100gm
- Silver Nitrate AR ACS
- Silver Sulphate SQ Qualigens
- Silver Nitrate Solution
- Silver Nitrate Merck 100g
- Silver Nitrate ER 25g
- Silver Nitrate for Analysis EMPARTA ACS
- Silver Nitrate 25g
- Silver Nitrate 100gm
- Silver Nitrate Acs 25g
- Silver Nitrate AR ACS
- Silver Nitrate for Analysis EMPARTA ACS (2)
- Silver Nitrate for Analysis EMPARTA ACS
- Silver Nitrate Merck 100g Merck
- Silver Nitrate Merck
- Silver Nitrate N10 C V S Qualigens
- Silver Nitrate Solution
- Silver Sulphate Ar Loba
- Silver Sulphate SQ Qualigens
- Laboratory Chemicals And Reagents
- Whatman Filter Paper
- Whatman Laboratory Filter Paper
- Whatman Filter Paper 127MM Grade 40
- Whatman Grade 41 Circles 125 mm
- Whatman 1822-037 Grade GF-C Glass Microfiber Filter Papers 1.2 um 37 mm
- Whatman Grade 1 Qualitative Filter Paper Standard 185 mm Circle 100 pack 1001-185
- Whatman Qualitative Filter Paper Grade 4
- Thimple HPGF 19X90MM 25-PK
- Laboratory Filter Paper
- Thimple HPGF 19X90MM 25- PK
- Whatman Filter Paper Grade 42 125mm
- Laboratory Filter Paper
- Thimple HPGF 19X90MM 25PK
- Whatman Filter Paper No 1, 125 mm
- Whatman 1822-025 Grade GFC Glass Microfiber Filter Papers 1.2 um 25 mm
- Whatman 1822-037 Grade GFC Glass Microfiber Filter Papers 1.2 um 37 mm
- Whatman Filter Paper 127MM Grade 40
- Whatman Filter Paper 1001-125 GR 1 12.5CM
- Whatman Filter Paper Grade 5 110MM Diameter
- Whatman Filter Paper Grade 41 125mm
- Whatman Filter Paper No 1, 110 mm
- Whatman Filter Paper No 1, 125 mm
- Whatman Grade 1 Qualitative Filter Paper Standard 185 mm Circle 100 pack 1001-185
- Whatman Grade 41 Circles 125 mm
- Whatman Grade GFC Glass Microfiber Filter Papers 47 mm
- Whatman High-Purity Glass Soxhlet Extraction Thimbles
- Whatman Laboratory Filter Paper
- Whatman Qualitative Filter Paper Grade 4 ,125mm
- Whatman Qualitative Filter Paper Grade 4
- Laboratory Plasticware
- Spinwin Conical Bottom Tube
- TARSONS SPINWIN Tube Conical Bottom PP with HDPE Closure 500041
- Spinwin Conical Bottom Tube
- Burette stand with Fisher Clamp (Plastic)
- Burette Transparent 50 mL Polylab
- Cryo Box PC Tarsons
- Laboratory Plastic Beakers
- Laboratory Plasticware
- Laboratory Tray PolyLab
- Magnetic Retriever PTFE ALNICO V MAGNET Tarsons
- Maxiamp PCR Tubes
- Measuring Cylinder 500 ml Polylab
- Micro centrifuge Tubes Pp Abdos
- Micro Tips Bulk
- Narrow Mouth Reagent Bottle - PP
- Polylab 250 ml Hexagonal Measuring Cylinder
- Reagent Bottle Wide Mouth 125ML
- Reagent Bottle Wide Mouth 250ml
- Reagent Bottle Wide Mouth 500ml
- Reagent Bottle Wide Mouth 1000ML
- Screw Cap Centrifuge Tube Round-Bottom, PP
- Spinwin Micro Centrifuge Tube
- Spinwin Tube Conical Bottom, PP with HDPE Closure
- Tarsons Measuring Beaker
- Tarsons Micro Tips 521010 2-200ul
- Tarsons Polypropylene Micro Tip
- TARSONS SPINWIN Tube Conical Bottom PP With HDPE Closure 500031
- TARSONS SPINWIN Tube Conical Bottom PP with HDPE Closure 500041
- TEST TUBE STAND (3 TIER) P.P
- Wash Bottle Ldpe Tarsons
- Honeywell Laboratory Reagent
- Sigma Aldrich Laboratory Reagent
- Himedia Laboratory Chemical
- Nutrient HiVeg Agar Himedia
- S Selective Supplement Himedia
- Alberts Stain A Laboratory Chemical
- U40 Supplement Himedia
- Sabouraud Dextrose Agar, Granulated Himedia
- Casein Soyabean Digest Broth (Soyabean Casein Digest Medium)
- Collagenase Type IV Solution
- LP Enrichment Supplement 1 Himedia
- MYP Agar Base Phenol Red Egg Yolk Polymyxin Agar Base) Himedia
- Luria Bertani Agar Plate Himedia
- Himedia Laboratory Reagent
- Pseudomonas Agar Base Powder M085
- Nutrient Agar Himedia
- Macconkey Agar Himedia
- Motility Test Medium HIMEDIA
- Antibiotic Assay Medium No 9 Polymyxin Base Agar
- Nutrient Broth Himedia
- Deoxycholate Citrate Agar Himedia
- Voriconazole VRC 30ST
- CLED Agar Bromo Thymol Blue HIMEDIA
- Chromogenic Coliform Medium W-Sterile Membrane Filter
- Chromogenic Coliform Medium W-Sterile Membrane
- Water Testing Instrument
- Industrial Chemicals
- Lab Chemical
- Laboratory Reagent
- Himedia
- AK Agar No2 Sporulating Agar
- Violet Red Bile Agar Himedia
- Antibiotic Assay Medium No 1 Seed Agar
- Antibiotic Assay Medium No 2 Base Agar
- Antibiotic Assay Medium No 9 Polymyxin Base Agar
- Antibiotic Assay Medium No 11
- Antibiotic Assay Medium No 35 (2)
- Antibiotic Assay Medium No 35
- Antibiotic Assay Medium No 40
- Barritt Reagent B (for VP test) Himedia
- Blood Agar Base 500g Himedia M073
- Buffered Sodium Chloride- Peptone Solution pH 7.0
- CDTA Monohydrate Hi AR ACS
- CDTA monohydrate Himedia
- Cedarwood Oil For Microscopy Optically Clear
- Cefepime 30mcg 1Vl Himedia (2)
- Cefepime 30mcg 1Vl Himedia
- Cetrimide Agar 500gm
- Cetrimide Agar Himedia 500ml
- Chromogenic Coliform Agar CCA Agar
- Chromogenic Coliform Medium WSterile Membrane Filter
- CLED Agar Bromo Thymol Blue HIMEDIA
- Columbia Blood Agar Base HIMEDIA
- Dey Engley Neutralizing Broth
- Diagnostic Stuart s Urea Broth Base
- EMB Agar Himedia
- Fluid Casein Digest Soya Lecithin Medium
- Fluid Thioglycollate Medium Thioglycollate Medium Fluid
- Glycine Hi-A ACS
- GN Broth Medium 11
- Gordon-McLeod Reagent Oxidase reagent
- Harris Haematoxylin
- HiCrome Candida Differential Agar
- Kovacs Indole Reagent
- Lauryl Sulphate Broth
- Lithium carbonate Hi AR ACS
- Luria Bertani Broth Miller
- Lysine Iron Agar HIMEDIA
- Lysine Iron Cystine Broth Base
- Macconkey Agar Himedia
- Macconkey Agar Srl
- MacConkey Agar wo CV, NaCl w 0.5% Sodium Taurocholate
- MacConkey Broth 500g
- Mannitol Salt Agar 500g
- Mannitol Salt Agar Base Himedia
- Mueller Hinton Agar Powder
- Mueller Hinton Agar
- Nutrient Agar Himedia
- Nutrient Broth Himedia
- Nutrient HiVeg Agar Himedia
- Paraffin Liquid Light
- Peptone Bacteriological Himedia
- Plate Count Agar Standard Methods Agar (2)
- Plate Count Agar Standard Methods Agar
- Pseudomonas Agar Base
- R2A Agar Himedia (2)
- R2A Agar HIMEDIA (3)
- R2A Agar Himedia (4)
- R2A Agar HIMEDIA
- Rappaport Vassiliadis Salmonella Enrichment Broth
- Reinforced Clostridial Agar
- Reinforced Medium for Clostridia
- Sabouraud Chloramphenicol Agar
- Sabouraud Dextrose Agar Himedia
- Sabouraud Dextrose Broth
- Safranin 05 wv
- Soyabean Casein Digest Medium Tryptone Soya Broth
- Soyabean HiVe Medium Sterile Powder Tryptone Soya HiVe Broth Sterile Powder)
- Soyabean HiVeg Medium Sterile Powder
- Soybean Casein Digest Agar
- Soybean Casein Digest Medium
- Sterile Cotton Swab
- Sterile Disposable L Spreader
- Sterile Disposable Petri Plates PW001
- Triple Sugar Iron Agar
- Violet Red Bile Glucose Agar
- Vogel Johnson Agar Medium
- Xylose Lysine Deoxycholate Agar XLD Agar
- Himedia Vails
- Itraconazole ITR 32mcg
- Bacitracin Himedia
- Acyclovir Plant Culture Tested Himedia
- Amikacin 10mcg 1Vl Himedia
- Amikacin AK 30mcg
- Amoxicillin AMX 1Vl
- Amoxyclav AMC 1Vl
- Amoxyclav Amoxicillin Clavulanic acid AMC 30 mcg
- Amphotericin B 32mcg
- Ampicillin 10mcg Himedia
- Ampicillin Cloxacillin Himedia
- Ampicillin Sulbactam 10 mcg
- Azithromycin 15mcg 1Vl Himedia
- Aztreonam AT 30 mcg
- Barritt Reagent A (for VP test) Himedia
- BD Difc Salmonella O Antis Poly Grp A I & Vi Lyo
- Bile Esculin Himedia
- Caspofungin CAS 32mcg
- Cefixime 5mcg 1Vl Himedia
- Cefotaxime Cephotaxime 30mcg Himedia
- Cefotaxime Clavulanic acid Himedia
- Cefoxitin Cephoxitin 30mcg 1Vl Himedia
- Ceftazidime 30mcg 1Vl Himedia
- Ceftriaxone 30mcg 1Vl Himedia
- Cefuroxime CXM 30 mcg
- Chloramphenicol 30mcg 1Vl Himedia
- Ciprofloxacin 5mcg 1VL Himedia
- Clindamycin 2mcg 1VL Himedia
- Co Trimoxazole Sulpha Trimethoprim Himedia
- Coagulase Plasma 01gm per vial
- Colistin Methane Sulphonate CL 10mcg
- Doxycycline Vails Himedia
- Erythromycin 15mcg 1 VL Himedia
- Fluconazole FLC 256mcg
- Fosfomycin Himedia
- Fraser Selective Supplement Himedia
- Fraser Supplement Himedia
- Gentamicin 10mcg VL Himedia
- Imipenem 10mcg Himedia
- Levofloxacin 5Mcg 1Vl Himedia
- Linezolid 30mcg 1VL Himedia
- LP Enrichment Supplement 1 Himedia
- Meropenem 10mcg 1VL Himedia
- Minocycline HIMEDIA
- Netillin Netilmicin Sulphate Himedia
- Nichrome Loop-D-2 Himedia
- Nitrofurantoin 300mcg 1VL Himedia
- Norfloxacin 10mcg 1Vl Himedia
- OA Selective Supplement Himedia
- Ofloxacin OF 5 mcg
- Optochin 5 mcg 50 discs vl Himedia
- Oxidase Discs 50 discs vl
- Oxidase Discs Himedia
- PALCAM Selective Supplement Himedia
- Piperacillin Tazobactam Himedia
- PTe 3.5 Selective Supplemen 1 ml per vial Himedia
- PYR Reagent Himedia
- Teicoplanin TEI 30 mcg
- Tetracycline 30 mcg 1VL Himedia
- Tetracycline TE 30 mcg
- Tigecycline 15mcg 1Vl Himedia
- Tobramycin TOB 10mcg
- U40 Supplement 5 ml per vial Himedia
- Vancomycin 30mcg Himedia
- Vancomycin Ezy MI Strip
- Voriconazole VRC 30ST
- Membrane Filters Milipore
- PVDF Membrane Filter 0.22 um Pore Size Millipore
- HVLP04700 Durapure PVDF.45UM WH PL 47MM
- Blue Membrane Filters Pore Size 045 Micron Diameter 47 Mm
- MCE .45UM WH PL 47MM 100PK
- MCE Membrane Filter 0.22 um Pore Size
- HAWP04700 Membrane Filter 0.45m 47mm diamter
- MCE Membrane Filter 1.2 Um Pore Size Millipore
- MCE Membrane Filter 5.0 um Pore Size
- MCE Membrane Filter, 0.22 um Pore Size Merck
- MF Millipore Membrane Filter 5 um pore size SMWP04700 Diameter 47mm
- Millipore Filter Papers
- Millipore Membrane Filter - 0.45 um pore size 47mm diameter
- PVDF Membrane Filter 0.45 um Pore Size HVLP04700
- SMWP04700 MILLIPORE Filter paper
- Water Testing
- Laboratory Chemical
- Potassium Iodide ER 250G
- Hydrogen Peroxide solution ER 500ML
- EDTA N50 Solution 500ML
- Rankem Benzaldehyde Reagent , Purity: 99%
- Lead Acetate Trihydrate SQ 500G
- SODIUM ACETATE TRIHYDRATE AR
- Potassium Dichromate SQ 500G Qualigens
- POTASSIUM NITRATE AR 500G (meets analytical sp
- Acetic Acid Glacial 500ml
- Acetonitrile Gradient Grade 2.5l
- Ammonia Buffer Solution 500ML Qualigens
- Ammonium Bifluoride SQ 500G
- Potassium Sulphate 500gm
- Ammonium Ferrous Sulphate 500gm
- Bacillocid Extra 5ltr
- BD BBL IsoVitaleX Enrichment
- Benzol Peroxide Loba
- Boric Acid Crystals LR
- Bromopropylate Sigma Aldrich
- Buffer Solution PH 4.0 550ml Qualigens
- Buffer Solution PH 7.0 550ml
- Buffer Solution Traceable T Ph.4
- Calcium Chloride Dihydrate ER 500g
- Calcium Chloride Fused SQ 500G
- Chlorbenzene Rankem 500ml
- Cyclohexane SQ Qualigens
- Dextrose Anhydrous SQ 500g
- Di Sodium Tetraborate
- Diethyl Ether SQ Qualigens
- Disposable Forceps Himedia
- Dithizone 85% Extra Pure Loba
- EC Broth Himedia
- Ethylene Glycol 500ml
- Ferrous Sulphate Heptahydrate Reagent
- Gadolinium III Sulphate Octahydrate 99.9%
- Hematoxylin Harris
- HEXAMINE Methenamine Hexamethylene Tetramine HMTA
- Hexane ER 500ML
- Hydrochloric Acid SQ 5l
- Hydrochloric Acid SQ Qualigens
- Hypersil BDS C8 HPLC Columns
- Iodine Resublimed SQ 100g
- Labolene 5ltr Qualigens
- Loba Chemie Bromine Water Approx. 0.2 M Loba
- Lovibond Dpd Tablets No 4 For Water Testing
- M Chloroaniline MCA Pure 99%
- Manganese II Chloride Tetrahydrate Extrapure 500G
- Manganese Standard Solution 1000 ppm BND CRM
- Merck Acetone EMPLURA 2.5l Lr Grade
- Mercury II Sulfate EMPLURA
- Methyl Red Indicator Solution 125ML Qualigens
- Nitric Acid About 69 For Analysis EMPARTA AC
- Nitrite Standard Solution 1000 ppm BND CRM
- Orthophosphoric Acid 88 AR 500ml
- Orthophosphoric Acid Sp Gr 1.75 AR Phosphoric Acid AR
- Phosphorus AAS Standard Solution LOBA
- Potassium Acetate For Molecular Biology 99.5%
- Potassium Chloride 500gm Qualigens
- Potassium Ferrocyanide SQ QUALIGENS
- Potassium Hydrogen Phthalate ER ACS Qualigens
- Potassium hydroxide Plant Culture Tested Himedia
- Potassium Iodide SQ Qualigens
- Potassium Permanganate AR ACS Complies USP
- Potassium permanganate EMPLURA MERCK
- Rankem Ammonia Solution
- Salicylic Acid AR
- Sodium Bisulphite SQ 500g Qualigens
- Sodium Dihydrogen Ortho phosphate Dihydrate SQ
- Sodium Hydroxide Pellets SQ 500g Qualigens
- Sodium Phosphate Dibasic Dodecahydrate Extrapure AR 99
- Spectroquant Lead Test Range
- Stannous Chloride SQ 100G Qualigens
- Styrene Monomer Extapure 99.5%
- Sulfanilic Acid 25g
- Sulfuric Acid About 98% Emplura
- Thymolphathalin Indicator Solution 125ml Qualigens
- Turbidity 100 NTU Calibration Standard
- Universal Indicator PH 4 11 500ML
- Virosil Pharma Disinfectant
- Paraffin Liquid Light
- Ammonium Dihydrogen Orthophosphate 500gm
- Aniline Laboratory Reagent
- Bhi Broth Himedia
- EDTA Disodium Salt SQ 500g Qualigens
- Erichrome Black T 25G
- Fluorescein diacetate Sigma
- Hydrochloric acid about 37 Emparta Merck
- Hydrochloric Acid Rankem
- Hydroxylammonium chloride SQ 500g
- Lovibond Dpd Tablets No 1 For Water Testing
- Methanol Specially Dried 99%
- Nitric Acid ER 2.5l
- N-Methyl-2-Pyrrolidone NMP ACS 99%
- OA Selective Supplement Himedia
- Orthophosphoric Acid SQ 500ml
- Periodic Acid SQ Qualigens
- Poly Acrylic Acid Sodium Salt
- Poly Tetramethylene Ether Glycol 1000
- Potassium Iodide 100gm
- Potassium Persulphate ER 500G
- QuickRxn XhoI 20ul
- Silicon standard solution
- Sodium Acetate Anhydrous SQ 500g Qualigens
- Sodium Acetate Trihydrate ER 500g Qualigens
- Sodium Phosphate Monobasic Dihydrate For Molecular Biology 99%
- SRL Water Nuclease Free
- Tetraisopropyl Methylenediphosphonate TCI
- Water Nuclease Free Srl
- Perchloric Acid In Anhydrous Acetic Acid
- 2-Chlorotoluene TCI
- Acetone ER Qualigens
- Nitric Acid ER 500ML
- SODIUM SULPHIDE HYDRATE FLAKES IRON FREE
- Sodium Thiosulphate ER 500G
- Mercuric Sulphate 100G
- Handwash savlon professional white Can 5L
- Acetic Acid Glacial
- Hypersil GOLD C18 Selectivity HPLC Columns
- Buffer Tablets PH 7.0
- Buffer Tablets PH 9.2 Qualigens
- Calcium Hardness Tablets
- Chlorotex Reagent 100ml
- Distilled Water SQ 5l
- Ethyl Acetate SQ 2.5L
- Iso Propyl Alcohol SQ
- Peptone Bacteriological Himedia
- TDS 442 tandard Solutions 300ppm
- Qualigens N Butyl Alcohol ER
- Laboratory Chemicals
- Contact Us